Theme
Immune-checkpoint inhibitors and CAR-T cell therapy opened the era of immuno-regulatory therapy. We are trying to develop innovative immuno-regulatory therapies against various diseases such as cancer and autoimmune diseases based on the understanding of the regulatory mechanisms of immune system.
About Research
Elucidating the regulatory mechanisms of immune system
Immune checkpoint inhibitors that antagonize the inhibitory co-receptors PD-1 and CTLA-4 improved the outcomes of diverse cancer types and revolutionized cancer treatment. However, response rates are rather limited and immune-related adverse events (irAEs) have also been observed in human patients receiving these inhibitors. This is indicative of the continued need to decipher the complex biology of inhibitory co-receptors to increase response rates and prevent such unwanted side effects in cancer patients.
Stimulatory and inhibitory co-receptors tightly control the activation of lymphocytes by regulating the quality and the quantity of the antigen receptor signaling to optimize beneficial immune responses while avoiding autoimmunity and excess immune responses. In addition to PD-1 and CTLA-4, many other co-receptors have been identified and regarded as potential drug targets. Although these co-receptors are supposed to have unique function and cooperate each other, their functional differences and coordination remain to be clarified. The primary aim of our laboratory is to elucidate the molecular and cellular mechanisms of PD-1 as well as other co-receptors in the regulation of immune responses. By fully understanding the regulatory mechanisms of immune system, we believe we can manipulate immune responses precisely to control various diseases such as cancer and autoimmune diseases.
Publication
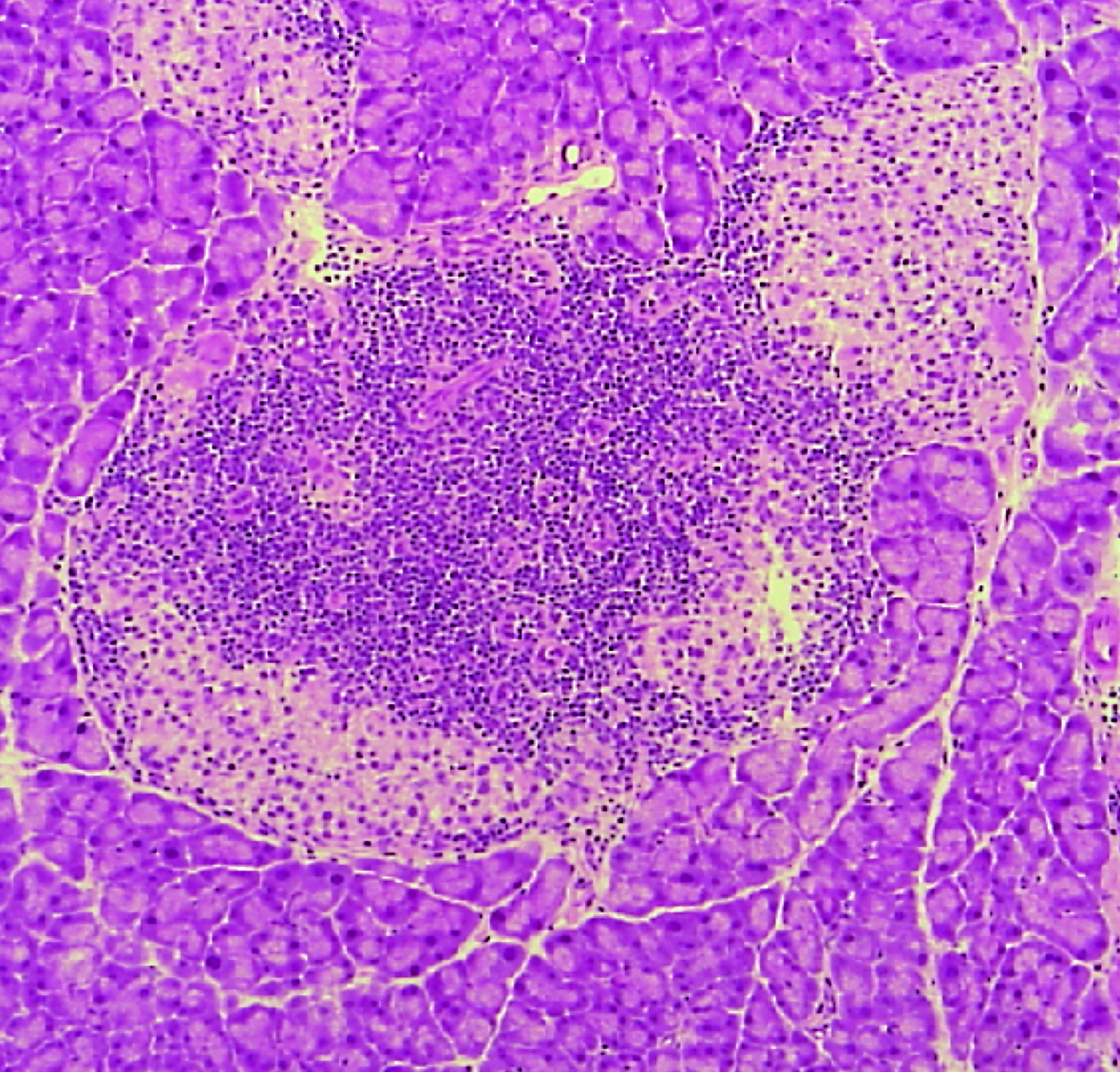
- Binding of LAG-3 to stable peptide-MHC class II limits T cell function and suppresses autoimmunity and anti-cancer immunity.
Maruhashi T, Sugiura D, Okazaki IM, Shimizu K, Maeda TK, Ikubo J, Yoshikawa H, Naenaka K, Ishimaru N, Kosako H, Takemoto T, and Okazaki T.
Immunity, 55:912-924.e8, 2022.
- PD-1 agonism by anti-CD80 inhibits T cell activation and alleviates autoimmunity.
Sugiura D, Okazaki IM, Maeda TK, Maruhashi T, Shimizu K, Arakaki R, Takemoto T, Ishimaru N, and Okazaki T.
Nat Immunol, 23:399-410, 2022.
- T cell-interinsic and -extrinsic regulation of PD-1 function.
Sugiura D, Shimizu K, Maruhashi T, Okazaki IM, and Okazaki T.
Int Immunol, 33:693-698, 2021.
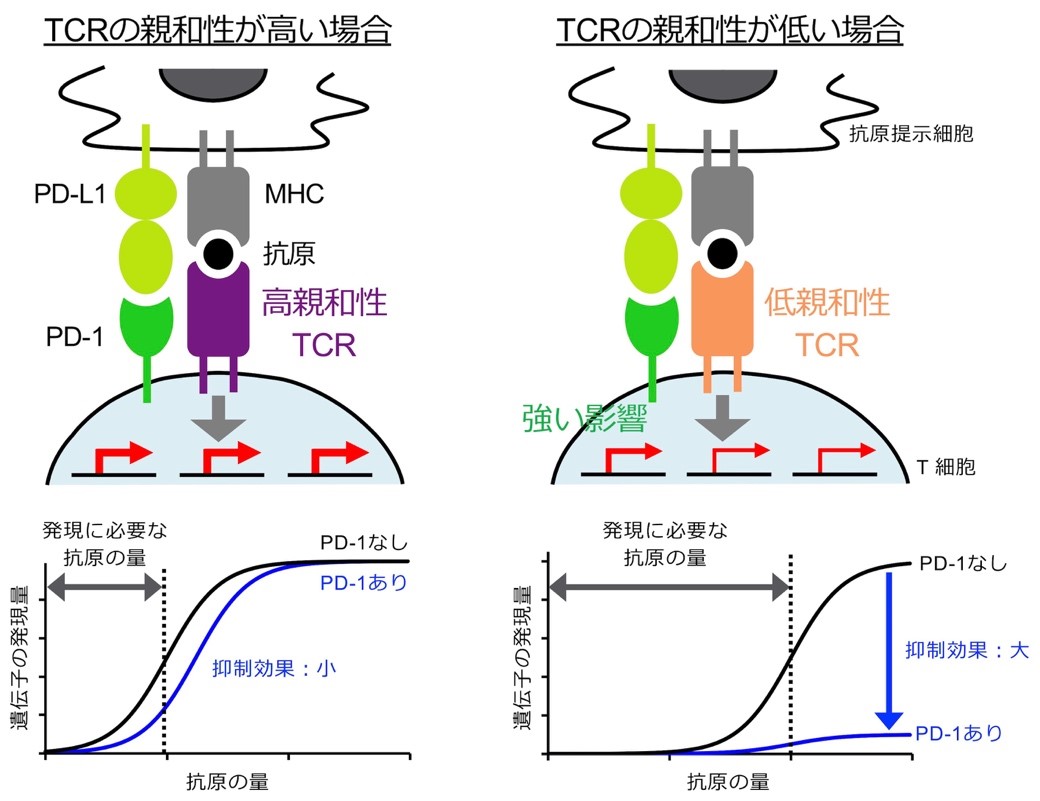
- PD-1 preferentially inhibits the activation of low affinity T cells.
Shimizu K, Sugiura D, Okazaki IM, Maruhashi T, Takemoto T, and Okazaki T.
Proc Natl Acad Sci USA, 118:e2107141118, 2021.
- PD-1 imposes qualitative control of cellular transcriptomes in response to T cell activation.
Shimizu K, Sugiura D, Okazaki IM, Maruhashi T, Takegami Y, Cheng C, Ozaki S, and Okazaki T.
Mol Cell, 77:937-950.e6, 2020.
- LAG-3: from molecular functions to clinical applications.
Maruhashi T, Sugiura D, Okazaki IM, and Okazaki T.
J Immunother Cancer, 8:e001014, 2020.
- Restriction of PD-1 function by cis-PD-L1/CD80 interactions is required for optimal T cell responses.
Sugiura D, Maruhashi T, Okazaki IM, Shimizu K, Maeda TK, Takemoto T, and Okazaki T.
Science, 364:558-566, 2019.
- PD-1 primarily targets TCR signal in the inhibition of functional T cell activation.
Mizuno R, Sugiura D, Shimizu K, Maruhashi T, Watada M, Okazaki IM, and Okazaki T.
Front Immunol, 10:630, 2019.
- LAG-3 inhibits the activation of CD4+ T cells that recognize stable pMHCII through its conformation-dependent recognition of pMHCII.
Maruhashi T, Okazaki IM, Sugiura D, Takahashi S, Maeda TK, Shimizu K, and Okazaki T.
Nat Immunol, 19:1415-1426, 2018.
- PD-1 and PD-1 ligands: from discovery to clinical application.
Okazaki T and Honjo T.
Int Immunol, 19(7): 813-824, 2007.
Taku Okazaki
Professor
Ph.D.
Graduate School of Medicine
Il-mi Okazaki
Associate Professor
Ph.D.
Graduate School of Medicine
Daisuke Sugiura
Associate Professor
Ph.D.
Graduate School of Medicine
Takumi Maruhashi
Research Associate
Ph.D.
Kenji Shimizu
Research Associate
Ph.D.
Junko Tsueda
Technical Specialist
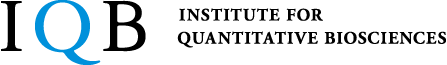

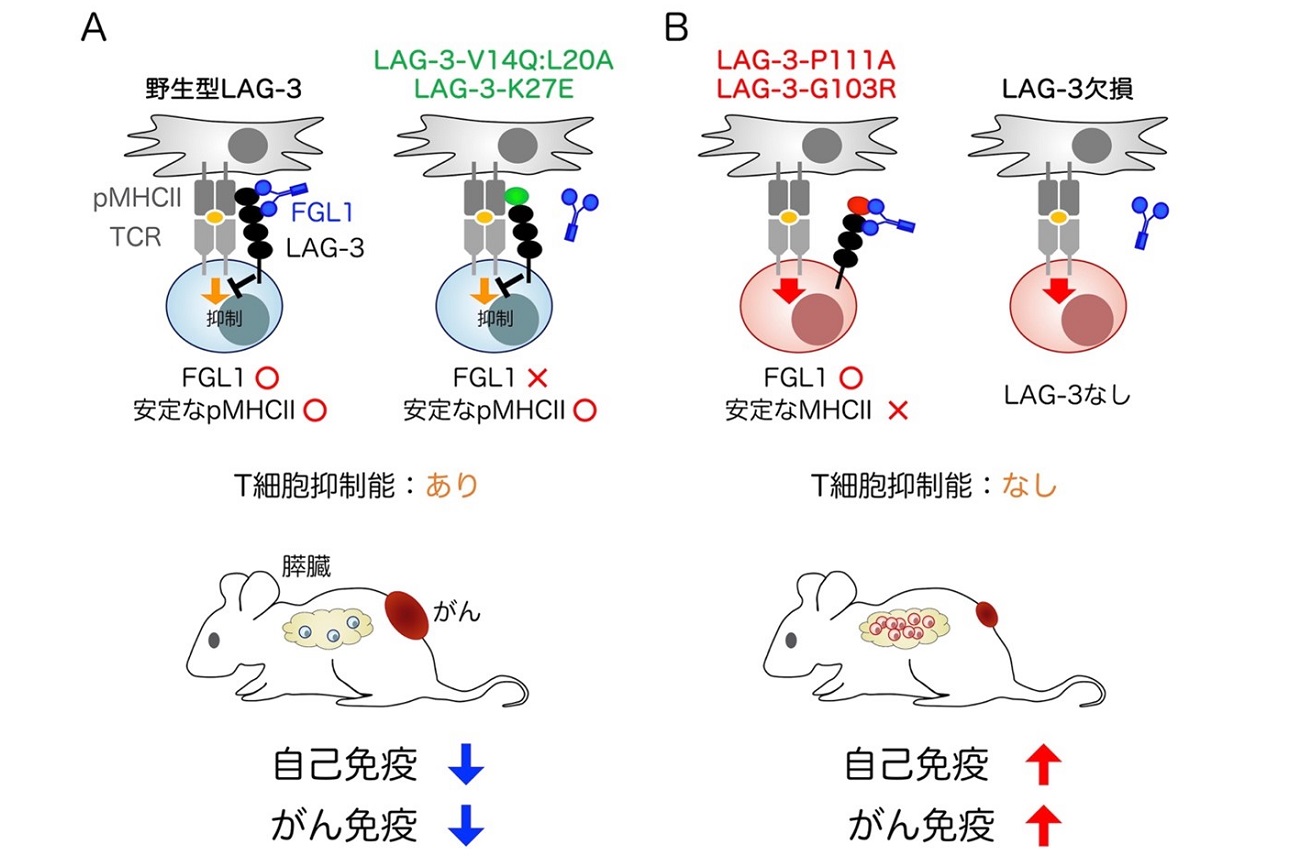






B:PD-1 inhibits T cell activation upon associating with PD-L1 (left). PD-1 fails to associate with PD-L1 when PD-L1 forms cis-duplex with CD80 (middle) (Sugiura et al, Science, 2019). Liberation of PD-L1 from cis-PD-L1/CD80 duplex using anti-CD80 Ab allows PD-1 to associate with PD-L1, inhibit T cell activation, and alleviate autoimmunity (right) (Sugiura et al, Nat Immunol, 2022).